Intragastric Balloon Therapy for Weight Loss
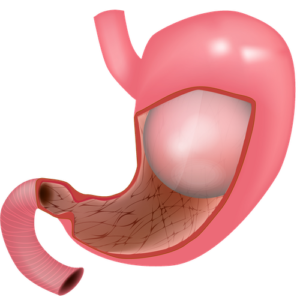
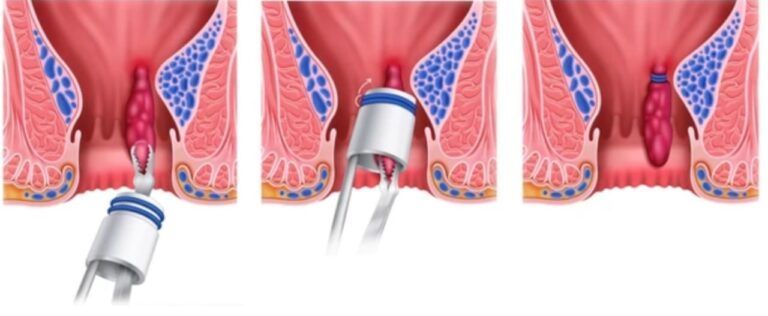
Intragastric balloon therapy is an endoscopic and temporary method of inducing weight loss. It entails placing a soft saline- or gas-filled balloon in the stomach to promote a feeling of satiety and restriction. Because its weight loss mechanism is mostly restrictive, its efficacy largely relies upon a patient’s adherence to lifestyle changes (ie, diet and exercise).
- Intragastric balloon therapy is an option for the treatment of obesity in patients with a body mass index (BMI) of greater than 30 kg/m 2 in the United States. Patients must have failed previous attempts at weight management with diet and exercise alone. Intragastric balloon therapy can be used for overweight or obese patients who do not yet meet BMI criteria for surgery. In patients who are candidates for bariatric surgery, intragastric balloon therapy can be used as a bridge to surgery (when BMI ≥50 kg/m 2 ) or in place of surgery for those who refuse or who lack access to surgery.
- Absolute contraindications to intragastric balloon therapy include previous gastric surgery, coagulation disorders, any bleeding lesion in the gastrointestinal tract, pregnancy or desire to become pregnant, alcoholism or drug addiction, severe liver disease, and any contraindication to endoscopy. Relative contraindications include previous abdominal surgery, large hiatal hernia, inflammatory bowel disease, chronic nonsteroidal anti-inflammatory drug use, and psychiatric disorders.
- Intragastric balloons differ in their volume, duration in the stomach, adjustability, and means of insertion and removal:
- Orbera and ReShape are endoscopically inserted and removed balloons that remain in the stomach for six months. Both balloons have been approved by the US Food and Drug Administration (FDA).
- Obalon balloons are swallowed but endoscopically removed at six months; the aggregate volume varies depending upon the number of balloons inserted (one to three). Obalon balloons have also received FDA approval.
- The Spatz balloon is endoscopically inserted and removed; its volume can be adjusted while remaining in the stomach for up to one year.
- The Elipse is a procedureless balloon that does not require endoscopy for insertion or removal. At approximately four months, it deflates by itself and is naturally excreted.
- Intragastric balloon therapy can achieve 8 to 15 percent total body weight loss (or 25 to 38 percent excess weight loss) depending on the type of balloon used. Although there are no direct comparison trials, this degree of short-term weight loss compares favorably with behavior modification or pharmacotherapy but less favorably with bariatric surgery.
- During the first week of intragastric balloon therapy, a majority of patients will develop some form of gastrointestinal symptoms due to gastric accommodation to the balloon. The combination of a proton pump inhibitor, an antiemetic, and an anticholinergic helps most patients through the first week of therapy, after which time the symptoms greatly diminish. Serious adverse events (including death) can, but rarely, occur during intragastric balloon therapy and can be minimized by thorough patient education, closely monitoring for complications, timely balloon removal, and adequate training of endoscopists performing the procedure.
- Regaining of lost weight is a common problem in treating obesity. After intragastric balloon therapy, behavior modification (diet, exercise, healthy sleeps habits , lifestyle changes) should be continued to minimize subsequent weight gain. Additional adjunctive measures to sustain or augment weight loss after intragastric balloon therapy include weight loss medications, administered by a gastroenterologist , repeated balloon therapy after at least a one-month “balloon holiday,” balloon volume adjustment (eg, with Spatz), or bariatric surgery.
Source: Uptodate ®
In our weight loss clinic, we follow closely our patients after balloon placement. The patients see our registered dietitian often in order to learn how to modify their diet habits. In some cases, we help them achieve their goals by also adding FDA appetite suppressant medications into their regimen.
Learn More About Gastric Balloon And Gastric Sleeve
Benefits Of Orbera Gastric Balloon
A.G. Assoc. Recommends Intragastric Balloons for Obese Patients
Benefits Of The Gastric Sleeve
About the Gastric Balloon Procedure
Intragastric Balloon Therapy For Weight Loss
The post Intragastric Balloon Therapy for Weight Loss appeared first on Gastro SB.